Introduction to Copper: Types of Copper
Copper Applications in Metallurgy of Copper & Copper Alloys
High Conductivity Copper | Deoxidized Copper | Copper Alloys | Copper in Other Metals | Recycling of Copper | Health | Environment | Copper Compounds
High Conductivity Copper 
A third to one-half of all copper produced is used in some form for applications in electrical engineering and the supply of domestic electricity. The reason is simple — among the readily available engineering materials copper is unique. Not only is it extremely ductile and capable of being formed into a wide range of products — especially wire —with ease, but it has almost uniquely high values of thermal and electrical conductivity, exceeded only by silver. The high electrical conductivity is especially important for the efficient transmission and utilization of electrical energy, and copper is therefore the principal material for building wire and cable, busbars, and motor and transformer windings.
There are a number of types, or grades, of what is essentially pure copper. These grades differ slightly in purity and in the types of "impurity" elements contained, but all contain at least 99.3% Cu. Further information on this subject can be found later in this article. A listing of copper grades classified under the Unified Numbering System (UNS) is accessible by searching Properties of Wrought and Cast Copper Alloys database. The following are the popular types of wrought copper, each suitable for a variety of applications. A few comparable alloys of cast copper are also produced but will not be discussed here owing to the far greater commercial importance of the wrought alloys.
High conductivity (HC) electrolytically refined copper is used for most electrical applications such as wire and cable, busbars and windings. The most commonly used grade is known in North America as electrolytic tough pitch (ETP) copper, UNS C11000, and as 'electro' in other countries. ETP copper exhibits a nominal conductivity of 100% to 101.5% IACS (for International Annealed Copper Standard, the value of conductivity established about 100 years ago for the purest copper of the time). High conductivity copper is very readily worked, both hot and cold. It has excellent ductility, which means that it can be easily drawn to fine wire sizes, and it is available in all other fabricated forms. ETP copper contains a minimum of 99.90% copper, with oxygen being the principal secondary element.
There are several highly refined grades of copper that contain almost no oxygen or other impurities. These are the so-called oxygen-free high-conductivity coppers. (The acronym for the term, oxygen-free high conductivity copper, OFHCTM, is a registered trademark of Phelps Dodge Specialty Copper Products. Comparable generic products are generally referred to simply as OF copper.) OF copper is produced by casting electrolytically refined copper in a controlled, i.e., non-oxidizing atmosphere. It is used where ease of welding and brazing are especially important. OF coppers exhibit electrical conductivity above 100% IACS.
Certified oxygen-free high-conductivity copper (Electronic Grade, UNS C10100) contains a minimum of 99.99% Cu, making it the purest metal in common use. It contains very low levels of residual volatile impurities and is therefore used for high vacuum electronic applications such as transmitter tubes, waveguide tubes, linear accelerators and glass-to-metal seals. Virtual elimination of oxygen also avoids certain welding problems encountered in oxygen-bearing grades, thus improving fabricability.
 Figure 6. Free machining copper
Figure 6. Free machining copperFree-machining copper has an addition of around 0.5% of sulfur or tellurium which raises the machinability rating of copper from a value of 20 to 90 (based on a scale of 100 headed by free-cutting brass, UNS C36000). Lead acts similarly in copper, although free-cutting leaded coppers as such (i.e., other than special materials used for bearings) are not listed under current UNS classifications. Applications for such free-machining grades as tellurium-bearing coppers (UNS C14500 and other) and sulfur-bearing copper (C14700) include machined electrical components, gas-welding nozzles and torch tips and soldering iron tips.
The cost of these components for the electrical engineering industry is kept low by making them using a free-machining grade of copper. They are cast or forged near to final shape and finish machined to close tolerances. (Thomas Bolton)
Deoxidized Copper 
Deoxidized copper is used for the other major area of application of the coppers in building construction apart from electrical services, the principal uses being for central heating systems, tube for gas and water supply, and sheet for roofing and other architectural applications. The oxygen in copper is usually removed by the addition to the melt of phosphorus as a copper-phosphorus hardener, or boron in the case of castings. This gives a material that can readily be brazed or welded without fear of embrittlement through contact with hydrogen. It is therefore ideal for use in plumbing systems and domestic gas supply. The ability of copper to form a protective and aesthetically pleasing surface, or patina, by weathering has encouraged its use for roofing large buildings over many centuries. Today, such architectural uses are expanding to include wall panels, column cladding and other items.
Copper Alloys 
Copper forms alloys more freely than most metals and with a wide range of alloying elements. Zinc, tin, nickel and aluminum are the most common alloying additions and produce the following simple alloy types. There are several other types not listed here.
| Addition | Alloys |
|---|---|
| Tin | Bronze |
| Tin and Phosphorus | Phosphor bronze |
| Aluminum | Aluminum bronze |
| Zinc | Brass |
| Tin and Zinc | Red Brass or Gunmetal |
| Nickel | Copper-nickel |
| Nickel and zinc | Nickel silver |
In practice, many combinations of alloying elements are used in combination to optimize properties for a very wide range of uses. The effects of these are summarized in Figure 8.
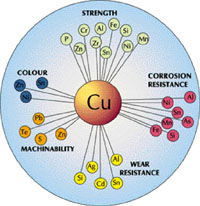 Figure 8. Some of the effects of alloying additions on the properties of copper
Figure 8. Some of the effects of alloying additions on the properties of copperAlloys based upon copper are classified as non-ferrous (ferrous materials are iron-base; for example, steel). Useful alloying additions of other elements to the alloy types listed above in small amounts can include aluminum, arsenic, antimony, beryllium, cadmium, chromium, cobalt, cadmium, iron, lead, manganese, nickel, niobium, oxygen, phosphorus, silicon, silver, sulfur, tellurium, tin, zinc and zirconium. All are found in standard coppers and copper alloys and are added as required in small amounts to give specific properties suitable for many demanding applications.
Some alloying elements have been in use with copper since early times. The development of metallurgical and corrosion science knowledge has provided many answers to specific metallurgical or corrosion phenomena, and these improvements, in turn, have at times lead to the use of other alloying elements with copper. A good example of this synergism is the development of new and improved alloys for use in the electronics industry.
Copper in Other Metals 
Apart from use in the copper-base alloys, there are other metals to which copper is added to improve properties. Structural steels can have an addition of about 0.5% copper to make them resistant to weathering and heavy progressive rusting. A few percent of copper also has a useful strengthening effect in steel.
The addition of copper (around 2 - 4 %) to duplex stainless steels and high-nickel super austenitic steels enhances corrosion resistance in acid environments and can also confer greater resistance to some forms of attack by seawater.
The most important alloy of nickel with copper, known as Monel metal, contains about 30-35% copper. It is highly resistant to many forms of corrosion, especially in chemical processing and marine applications.
Alloys of aluminum with about 4% of copper can be heat treated to give high strength. For example, copper is an essential constituent in the aluminum casting alloys used in automotive engine blocks and cylinder heads.
Recycling of Copper 
Copper and copper alloys have been recycled for thousands of years. (It has been said that even some of the copper used in ancient Egyptian plumbing tube has probably been recycled many times over the centuries.) This has been a normal economic practice stemming based in part on copper's high value. One of the wonders of the old world, the Colossus of Rhodes, Greece, an enormous statue spanning the entrance to Rhodes Harbor in Ancient Grecian times, was said to have been made of copper. No trace of it remained after it was recycled to make useful artifacts.
The entire economy of the copper and copper alloy industry is dependent on the economic recycling of any surplus products. Estimates place the amount of recycled copper at around 35% of the total annual copper consumption in the USA. Use of the most suitable and cheapest feedstock — scrap copper, for instance — for making components enables materials such as copper alloys to be manufactured at reasonable cost, while at the same time saving the energy needed to make virgin metal from ore.
Good quality high conductivity copper can be recycled by simple melting and check analysis before casting, either to finished shape or for subsequent fabrication. However, this normally only applies to premium quality process scrap (so-called prompt scrap) arising within a copper works. Copper that has been brazed, soldered welded or plated may have other metals present that make it unsuitable for re-use as high conductivity copper, but such copper can be — and usually is — remelted and used to make useful alloys. Where copper has been mixed with other metals and must be re-refined, it is normally remelted and cast to anode shape so that it can be electrolytically refined. In some cases, as with plumbing tube, it may simply be fire-refined in a suitable furnace. If, however, the level of impurities in the cast anode is significant, it is unlikely that the cathode produced will then meet the very high standards required of Grade 'A' Copper (the commercial designation for a type of cathode copper traded on exchanges) used for the production of fine wires. Such copper is used as feedstock for the production of alloys.
Where copper and copper alloy scrap is very contaminated and unsuitable for simple remelting, it can be recycled by other means to recover the copper either as the metal or to produce some of the many copper compounds essential for industry and agriculture. This is the usual practice for recovery of usable copper in slag, dross or mill scale arising from production processes or from life-expired assemblies of components containing useful quantities of copper.
Copper is never completely lost. Even when present in soil, copper is very slowly leached out and travels via the rivers to the sea, normally in the form of biologically unavailable chemical complexes or compounds. Here, over geologic periods of time, the copper may be precipitated out near centers of thermal activity and form nodules on the seabed. These can be extracted, although economics and political issues have prevented their exploitation thus far. In time they will form the basis for an ore bed that is pushed towards the surface by the earth's movement, and become a future source of valuable minerals.
Health 
Copper is one of a relatively small group of metallic elements that are essential to human health. These elements, along with amino and fatty acids as well as vitamins, are required for normal metabolic processes. However, as the body cannot synthesize copper, the human diet must supply regular amounts for absorption. Most meat, fish, shellfish and vegetables provide sources of copper, some more than others. (See Copper In Human Health, Quick Facts in Health & Nutrition and Copper Deficiency for further information.) The adult body contains between 0.6 and 0.95 milligrams of copper per pound of body weight (1.4 and 2.1 mg/kilogram). Hence, a healthy human weighing 130 pounds (60 kilograms) contains approximately 0.1g of copper. However, this small amount is absolutely essential to the overall human well-being; without it, one would surely die.
Copper combines with certain proteins to produce enzymes that act as catalysts to assist a number of body functions. Some of these enzymes help provide energy required by biochemical reactions; others are involved in the transformation of melanin for pigmentation of the skin and still others help to form cross-links in collagen and elastin and thereby maintain and repair connective tissues. This process is especially important for the heart and arteries. In fact, research suggests that copper deficiency is one factor leading to an increased risk of developing coronary heart disease.
Until recently, it was generally believed that most people consumed adequate quantities of copper. However, modern research has shown that this is not the case. Indeed, the World Health Organization recently noted that copper deficiency is likely a common and worldwide phenomenon. Many typical meals have been analyzed for their metals content. According to recent surveys conducted in the 1980's, only 75% of the U.S. population consumes the amount of copper per day estimated to be adequate by the US Food and Nutrition Board of the National Academy of Sciences.In the United Kingdom, it is now recommended that the daily intake should range from 0.4 mg/day for 1-3 year old children to 1.2 mg/day for adults.
Copper has been used as a medicine for thousands of years including the treatment of chest wounds and the purifying of drinking water (for further information on this topic, see Copper in My Medicine Chest? More recently, research has indicated that copper helps prevent inflammation in arthritis and similar autoimmune diseases. Research is going on into anti-ulcer and anti-inflammatory medicines containing copper, and its use in radiology and for treating convulsions and epilepsy. Although there is no epidemiological evidence that contact with copper can prevent arthritis, anecdotal claims have been made that the wearing of copper bracelets does alleviate symptoms. The article found at the URL at the beginning of this paragraph contains some further information on copper and arthritis.
Environment 
Copper is absolutely essential for the normal healthy growth and reproduction of most if not all higher plants and animals. The crop and animal losses caused by shortages of copper can, at one extreme, be total. For example, lambs can die of swayback, and crops on newly reclaimed peat bogs or sandy heathlands can fail completely. Fortunately, such occurrences are fairly rare. Far less dramatic, but economically very important, are the yield reductions of up to around 20% which can result from unrecognized (subclinical) copper deficiency in many crops without the appearance of any obvious symptoms. Even larger effects on live weight gain can occur in subclinically copper-deficient livestock, especially cattle. In addition to its essential role, copper has also been shown to have a very beneficial effect on the food conversion efficiency of pigs. Average daily live weight gains of up to 9.1% have been obtained by adding copper sulfate to the diet of fattening pigs.
Copper deficiency has been found throughout the world in all climatic zones where crops are grown or animals kept on farms. Its incidence varies according to soil, crop, livestock and management factors. In particular a deficiency can occur in crops growing on soils with a sandy texture (which drains away copper in water), on those rich in organic matter (which complex copper, making it biologically unavailable) and on calcareous soils (which tie up copper as a carbonate), but other soil factors can also cause a deficiency. Intensive management with highly productive crop cultivars and animal breeds can often exacerbate copper deficiencies, especially where much nitrogen and phosphorus fertilizer is used. Being such an important trace element, the distribution and concentration of copper in the environment is important. Typically there is 4.5 micrograms per gallon (1 µg/l) of copper in fresh water supplies.
In the ground, copper is normally present in compounds that are not easily soluble in water. Only a limited percentage, usually less than 1%, is available in soluble form and is therefore bioavailable. This copper can be taken up by the roots of plants as required and is then recycled as leaves and wood decay, concentrating in the top 4 inches (100 mm) or so of the soil. And as animals graze on these plants, copper in the soil is replenished from their excrement. Intensive farming without this recycling can lead to copper deficiency that has to be made up with fertilizer.
Copper Compounds 
Copper sulfate is commercially the most important copper compound, once called 'blue vitriol' from its close association with sulfuric acid. It is generally the starting stock for the manufacture of most other copper compounds. World consumption is around 200,000 tons per year, of which approximately 75% is used in agricultural applications. Cupric oxide, cuprous oxide, copper acetate, cupric chloride, copper oxychloride, cupric nitrate and copper napthenate are used selectively for these purposes for their ease of use or other special properties. Uses of copper compounds include:
- electrolyte for copper refining and electroplating
- anti-fouling paints
- catalysts for many industrial processes in the petrochemical and rubber industry and for textile manufacture.
- additives to cement for controlling setting rate and lichen growth
- addition as fungicide to plaster
- mordants for dyeing
- coloring agents for paints, glass and fireworks
- preservatives for paints, adhesives, timber, textiles and book bindings
Also in this Issue:
- Introduction to Copper: Applications
- Introduction to Copper: Types of Copper
- Introduction to Copper: Mining & Extraction
- Introduction to Copper: Fact Sheets
- Phelps Dodge Morenci Has Converted All Copper Production to Mine-for-Leach
- How Hydrometallurgy and the SX/EW Process Made Copper the "Green" Metal
- Introduction to Copper: Hot Links & Further Reading
